Molar mass of ag hydroxide8/15/2023  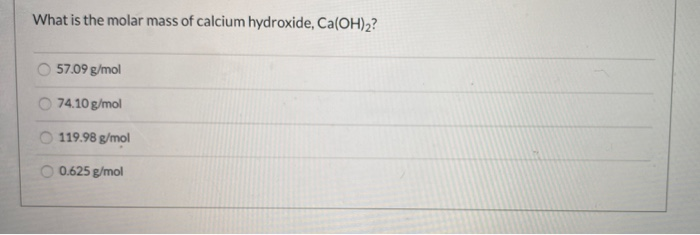
In aqueous solution both hydrogen and hydroxide ions are strongly solvated, with hydrogen bonds between oxygen and hydrogen atoms. It can also act as a Lewis base by donating a pair of electrons to a Lewis acid. In aqueous solution the hydroxide ion is a base in the Brønsted–Lowry sense as it can accept a proton from a Brønsted–Lowry acid to form a water molecule. 
Schematic representation of the bihydroxide ion pOH can be kept at a nearly constant value with various buffer solutions. For example, ammonia solutions have a pH greater than 7 due to the reaction NH 3 + H + ⇌ NH +Ĥ, which decreases the hydrogen cation concentration, which increases the hydroxide ion concentration. Addition of a base to water will reduce the hydrogen cation concentration and therefore increase the hydroxide ion concentration (increase pH, decrease pOH) even if the base does not itself contain hydroxide. The concentration of hydroxide ions can be expressed in terms of pOH, which is close to (14 − pH), so the pOH of pure water is also close to 7. The pH of a solution is equal to the decimal cologarithm of the hydrogen cation concentration the pH of pure water is close to 7 at ambient temperatures. Has a value close to 10 −14 at 25 ☌, so the concentration of hydroxide ions in pure water is close to 10 −7 mol∙dm −3, in order to satisfy the equal charge constraint. The equilibrium constant for this reaction, defined as The hydroxide ion is natural produced from water by the self-ionization reaction: H 3O + + OH − ⇌ 2H 2O Many inorganic substances which bear the word hydroxide in their names are not ionic compounds of the hydroxide ion, but covalent compounds which contain hydroxy groups. The corresponding covalently bound group –OH of atoms is the hydroxy group.īoth the hydroxide ion and hydroxy group are nucleophiles and can act as catalysts in organic chemistry. The corresponding electrically neutral compound HO Sodium hydroxide is a multi-million-ton per annum commodity chemical. The hydroxide ion forms salts, some of which dissociate in aqueous solution, liberating solvated hydroxide ions. It functions as a base, a ligand, a nucleophile, and a catalyst. It is an important but usually minor constituent of water. It consists of an oxygen and hydrogen atom held together by a single covalent bond, and carries a negative electric charge. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula.Hydroxide is a diatomic anion with chemical formula OH −. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.įinding molar mass starts with units of grams per mole (g/mol). 
These relative weights computed from the chemical equation are sometimes called equation weights. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. This site explains how to find molar mass. The reason is that the molar mass of the substance affects the conversion. To complete this calculation, you have to know what substance you are trying to convert. In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.Ī common request on this site is to convert grams to moles. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
